The time recorded is that taken for the liquid to flow through the capillary section of the viscometer, from one marker line to the other Where V = Kinematic Viscosity, C = Calibration constant of the viscometer, t = time. The unique constant of the capillary viscometer is also taken into account, with the final result calculated by the equation These all help to ensure more accurate results.Īfter equilibrating to the test temperature, the liquid is flowed through a capillary viscometer and the time is recorded. Some also have features to automatically calculate the viscosity, or even detect the results by optical sensors. Viscosity Baths are available in a range of specifications – models that heat, high temperature models for extended heating, or refrigerated models to run tests below room temperature. A viscosity bath is used to immerse capillary viscometers in liquid and hold them at a steady temperature – removing temperature as a variable in the test This means that accurately controlling the temperature of the liquid during testing is vital to ensure consistent results which can be compared across different batches or products.Ī viscosity bath is used to maintain samples at a steady and accurate temperature for Kinematic Viscosity testing to ensure that temperature is constant, and not a variable. You can see this effect in action by warming honey – at room temperature this is thick and slow to pour (high viscosity) but if you heat it in a pan you can see it becomes thin and fast to pour (low viscosity). 
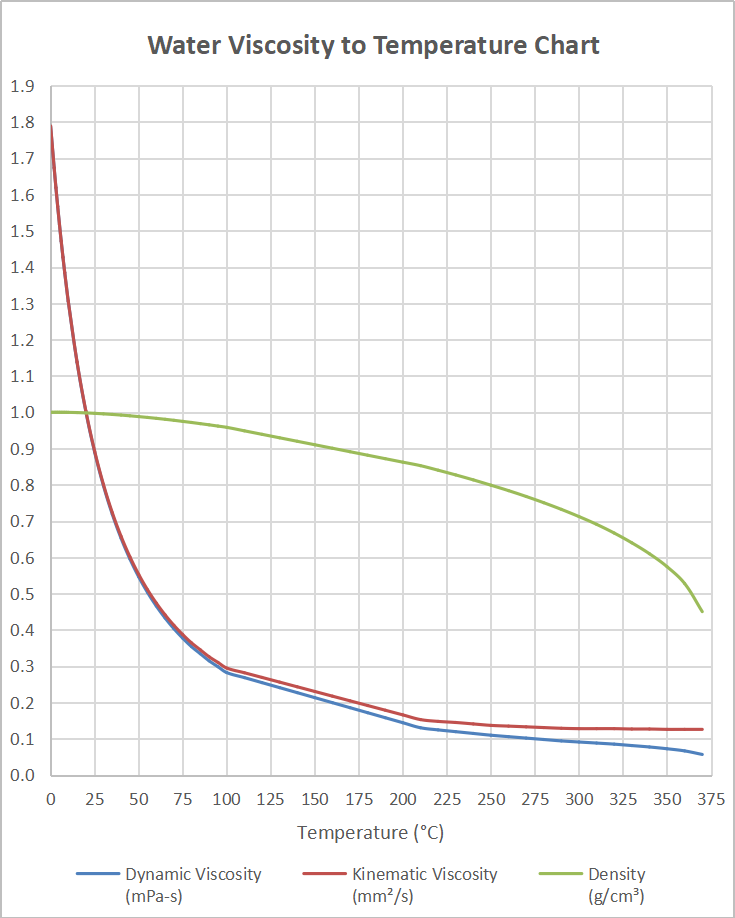
The impact of temperature on viscosity is huge – for every ☌ change, the viscosity can change by 2 to 10% (depending on the liquid). very thick liquid, slow to move when it is poured) very thin liquid, flows and pours fast) to high viscosity (i.e. The most commonly used unit is centiStokes (cSt) but some methods may report in mm 2/sīelow you can see the viscosity of some common liquids – ranging from low viscosities (i.e. What units is Kinematic Viscosity reported in? The time taken for the liquid to flow is measured – this could be the time it takes to travel through a capillary, or through a cup with a hole in the base. This is different to Dynamic viscosity, which measures the resistance of another object moving through the liquid. Kinematic Viscosity specifically is a measure of resistance to the liquid moving. Put simply, this means how thick or thin the liquid is – how easily it flows. The definition of viscosity is a measure of a liquid’s resistance to movement. We’ve explored some of the most commonly asked questions about viscosity testing what is kinematic viscosity? How is it measured? How does temperature affect viscosity? And how can you make sure your viscosity measurements are accurate? A glass capillary viscometer, used in Kinematic Viscosity measurements On the contrary, low viscosity fluids are more volatile, creating environmental problems.Kinematic Viscosity measurements are used in quality control and characterization tests in a wide range of fields. Oils with too high viscosity don't protect well against mechanical friction, resulting in heat generation. Viscosity will affect the thickness of the oil film as well as the effectiveness of its protective qualities. These films are used to protect equipment from mechanical wear and may also have extra benefits such as creating a barrier to corrosion. The viscosity of oil and lubricants are often measured in industrial settings. Kinematic viscometers determine the kinematic viscosity by measuring the time it takes for the fluid to pass through a portion of a capillary. Care must be taken when interpreting values and the temperature at which the values were measured. Understanding the distinction comes into play in industrial settings as record data is normally reported as kinematic viscosity, but onsite measurements are often measured as dynamic viscosity. Kinematic viscosity is not the same as dynamic viscosity, although they are related mathematically by the density of the fluid. This intermolecular friction is what makes the fluid resistant to change in shape and explains the thick and slow moving properties of highly viscous fluids. The stronger the intermolecular force, the more the molecules will stick together as they collide, manifesting a frictional interaction. Strongly polarized interactions like hydrogen bonding in water or interactions between large non-polar surfaces in viscous oils produce stronger intermolecular forces. Viscosity results from the intermolecular forces between molecules in a liquid. Corrosionpedia Explains Kinematic Viscosity
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
